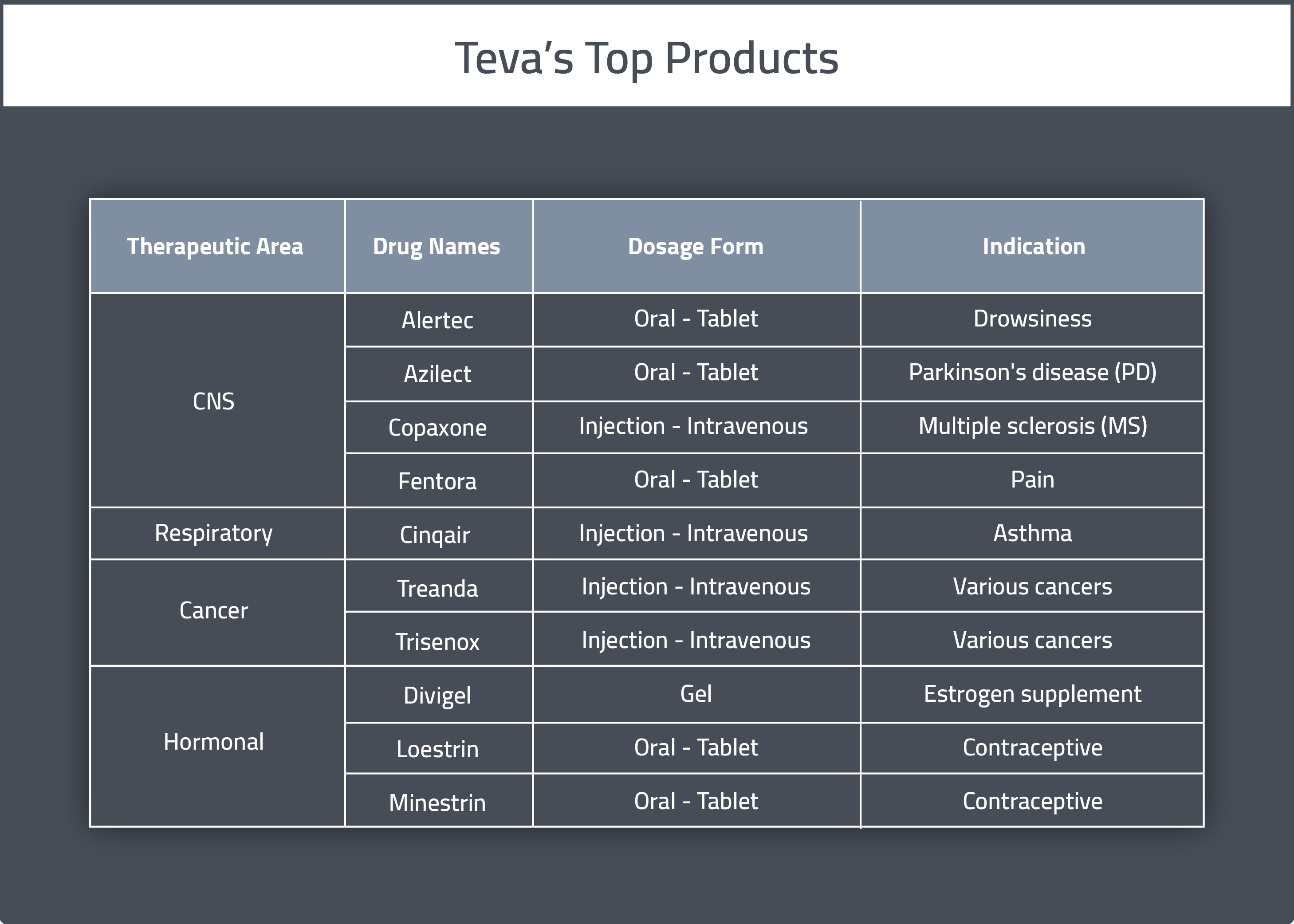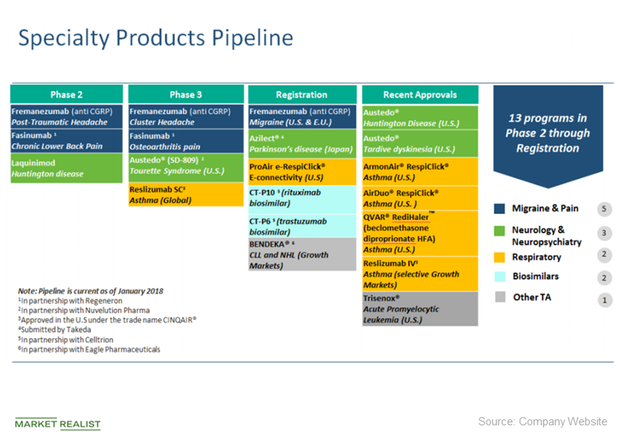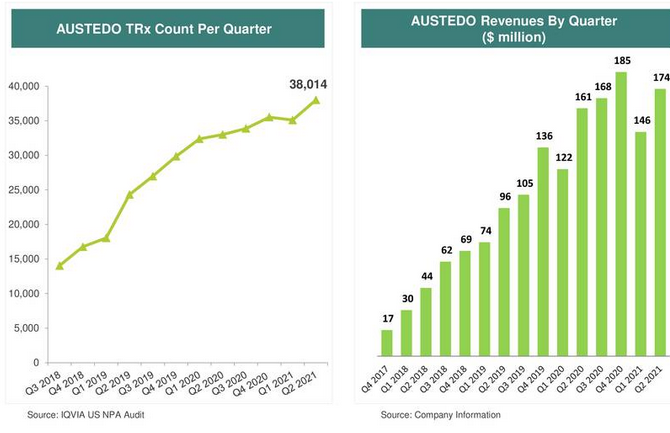
Teva Announces U.S. Approval of AJOVYTM (fremanezumab-vfrm) Injection, the First and Only Anti-CGRP Treatment with Both Quarterly and Monthly Dosing for the Preventive Treatment of Migraine in Adults ADDING MULTIMEDIA | BioSpace
Omeprazole Generic Drug Box by Teva Closeup Against White Editorial Image - Image of american, generics: 187803275