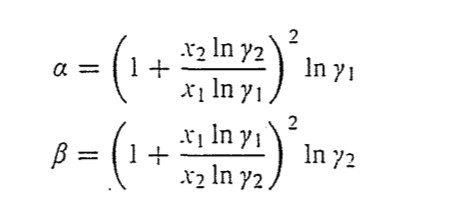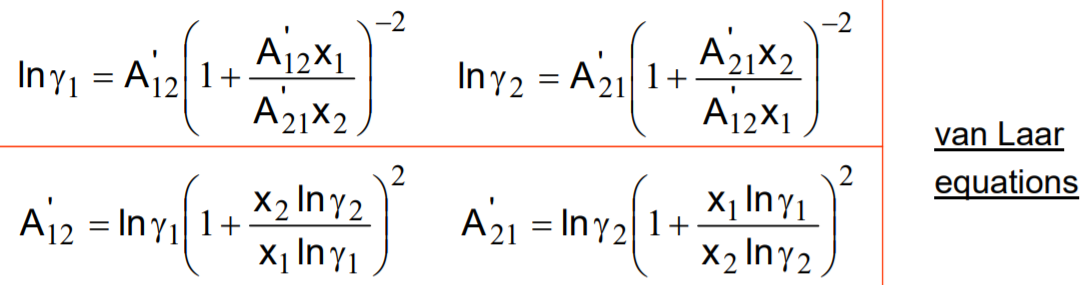DETERMINATION OF COMPOSITION-DEPENDENI' LIQUID ACTIVITY COEFFICIENrS1 BY USE OF THE VAN LAAR EQUATION GARRY LEONARD PIGG MASTER

Optimum binary interaction parameter and van Laar con- stants in the... | Download Scientific Diagram

SciELO - Brasil - Measurement and correlation of isobaric vapor-liquid equilibrium for the binary system of cyclopentane and tetrahydrofuran Measurement and correlation of isobaric vapor-liquid equilibrium for the binary system of cyclopentane

The Use of the van Laar‐Hildebrand Equation in High Polymer Chemistry - Staverman - 1948 - Bulletin des Sociétés Chimiques Belges - Wiley Online Library
![SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat](https://cdn.numerade.com/ask_images/1604588eda0f4ffc8897da2c6b3d4fee.jpg)
SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat
![PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar PDF] Extending the Van Laar Model to Multicomponent Systems~!2010-04-11~!2010-04-19~!2010-06-24~! | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0b4e610592ffe5443a53149e7d185fe8b3348cb2/5-Table3-1.png)
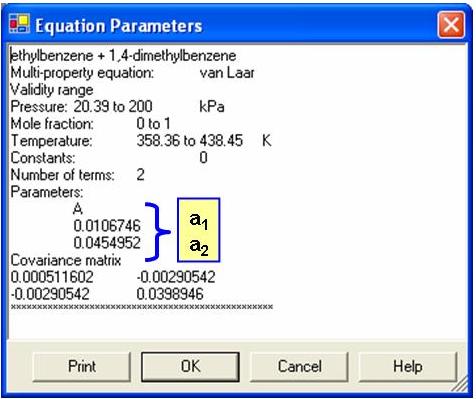








![2. Van Laar constants from solubility data [Stephen & Stephen, 75],... | Download Table 2. Van Laar constants from solubility data [Stephen & Stephen, 75],... | Download Table](https://www.researchgate.net/publication/319290742/figure/tbl5/AS:631676409491470@1527614800356/2-Van-Laar-constants-from-solubility-data-Stephen-Stephen-75-programme.png)